› Forums › Spectroscopy › Solar spectrum
- This topic has 6 replies, 4 voices, and was last updated 7 years, 11 months ago by
 Mr Jack Martin.
Mr Jack Martin.
-
AuthorPosts
-
16 June 2018 at 8:12 pm #574059
 Mr Jack MartinParticipant
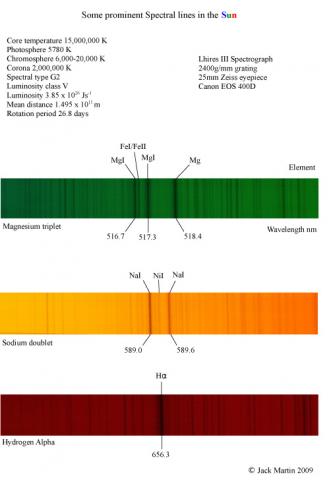
Mr Jack MartinParticipantWhy does sodium exist as a doublet and magnesium as a triplet in absorption lines of the Sun ?
Is this due to spin-spin coupling or the Zeeman effect ?
Regards,
Jack
Essex UK
Attachments:
18 June 2018 at 5:00 pm #579614 Robin LeadbeaterParticipant
Robin LeadbeaterParticipantHi Jack,
The two Na D lines are from the two different spin levels. (3s to 3p1/2 and 3p3/2 in absorption) There probably is Zeeman splitting of the lines as well but this will be very small in the solar magnetic field.
Cheers
Robin
18 June 2018 at 6:07 pm #579615 Thomas KillesteinSpectator
Thomas KillesteinSpectatorHi both,
Robin is correct, the splitting comes from the two 3p levels, which would be degenerate (i.e. no doublet) if not for the spin-orbit coupling – a sort of internal Zeeman effect from the magnetic field induced by the orbiting electron. This splits the levels and creates the doublet. If we apply a weak magnetic field (similar in size to the spin-orbit bit) we’ll get Zeeman splitting of these levels and end up in turn with 10 lines (5 from each line of the doublet)
The Na triplet is more complicated, but I think this comes from a similar 4s -> 3d transition, and selection rules exclude some of the 5 sublevels of the 3d orbital, giving a triplet – I’m slightly less clear on this one.
Regards,
Tom
18 June 2018 at 8:09 pm #579616 Dr Andrew SmithParticipant
Dr Andrew SmithParticipantIf you go on the NIST spectral data base and look at the relevant lines it shows you the transitions involved.
As above the splitting is due to the spin orbit coupling of the magnetic moments.
Regards Andrew
19 June 2018 at 8:47 pm #579620 Mr Jack MartinParticipant
Mr Jack MartinParticipantRobin, Thomas and Andrew,
Thank you for your answers, its become much clearer what’s going on and why.
Regards,
Jack
Essex UK
21 June 2018 at 6:49 pm #579633 Dr Andrew SmithParticipant
Dr Andrew SmithParticipantIt might be worth pointing out that the H alpha line is made up of 7 components with two being significantly stronger than the others. The splitting is small withthe largest spin orbit coupling split in the 2p level (J = 3/2 ,1/2)
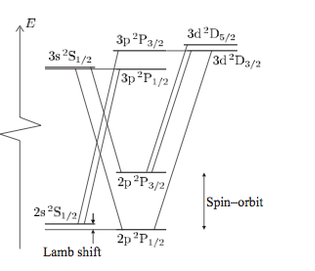
The theory of the spectrum of the Hydrogen atom traces the development of the quantum theory of light and matter. From the Bohr model where all the lines are degenerate in the prime quantum number (as the are in the Schrodinger model) through the development of Sommerfeld and Schrodinger to the relativistic theory of Dirac, Darwin, Lamb and on to QED for which Shin’ichirō Tomonaga, Julian Schwinger and Richard Feynman got the Nobel prize building on the work of many others, where they are all split apart.
21 June 2018 at 11:56 pm #579634 Mr Jack MartinParticipant
Mr Jack MartinParticipantAndrew,
Thanks for your input.
I never knew the h alpha line has 7 components.
Also a good point about the development of the quantum theory of light. No one person invents anything. In another example, Edison alone did not invent the light bulb, he needed Franklin’s work on electricity nearly 200 years before him.
Regards,
Jack
Essex UK
-
AuthorPosts
- You must be logged in to reply to this topic.
